In the past, the sweetener Stevioside continued to be subject of many controversial discussions. There are studies and expertises galore extensively discussing toxicity, carcinogenic effects and metabolism. Despite Stevia’s sweetness there remains a bitter aftertaste until its harmlessness is completely approved of.

Preface
Steviole glycosides are natural constituents of the plant Stevia rebaudiana, with at least 10 different types of glycosides found in its leaves, all differing in the residues of the basic Steviol-structure The main constituents are stevioside, rebaudioside A, rebaudioside C and dulcoside A. Other steviole derivates are negligible due to their low concentration. Depending on the cultivation conditions these constituents may vary from 4 to 20% of the dry matter. [1]
The sweetness of this plant is mainly due to stevioside, which is 250-300 times sweeter than glucose.
Acute and Chronic Toxicity
The toxicity and safety of stevioside was continually scrutinized in various scientific studies, the results of which do not allow any definite conclusions. Xili et al. calculated an ADI-value (acceptable daily intake) of 7,9mg stevioside/kg body weight. [4]
Stevioside shows, according to Toskulkao et al., a very low acute oral toxicity with mice, rats and hamsters. An LD50 of 8,2 and 17g/kg body weight was attested.
|
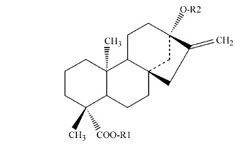 |
Fig. 1: Basic structure of steviol derivates according to Geuns [2]
|
Harmful effects are not attributed to stevioside itself, but to its residue steviole. Toskulkao et al. also prooved that the LD50 of steviole (90% purity) is between 5,2 and 6,1g/kg body weight with hamsters, and, with rats and mice, it is above 15g/kg body weight. Hamsters reacted more sensitively to steviole. [5]
Geuns’ studies on the chronic sub-acute toxicity suggest a higher NOEL than 2,5mg/kg bodyweight. This allows to deduce an ADI-value of 25mg/kg body weight with a safety factor of 100. [6]
Table1: Structures of stevioside and related compounds according to Geuns [3]]
|
Name of the Compound |
R1 |
R2 |
1 |
Steviol |
H |
H |
2
|
Steviolbioside |
H |
ß-Glc - ß-Glc (2 à
1) |
3
|
Stevioside |
ß-Glc |
ß-Glc - ß-Glc (2 à
1) |
4
|
Rebaudioside A |
ß-Glc |
ß-Glc - ß-Glc (2 à
1)
|
ß-Glc (3 à 1) |
5
|
Rebaudioside B |
H |
ß-Glc - ß-Glc (2 à
1)
|
ß-Glc (3 à 1) |
6
|
Rebaudioside C (Dulcosid
B) |
ß-Glc |
ß-Glc - a-Rha (2 à
1)
|
ß-Glc (3 à 1) |
7
|
Rebaudioside D |
ß-Glc - ß-Glc (2 à
1) |
ß-Glc - ß-Glc (2 à
1)
|
ß-Glc (3 à 1) |
8
|
Rebaudioside E |
ß-Glc - ß-Glc (2 à
1) |
ß-Glc - ß-Glc (2 à
1) |
9
|
Rebaudioside F |
ß-Glc |
ß-Glc - ß-Xyl
(2 à 1)
|
ß-Glc (3 à 1) |
10
|
Dulcoside A |
ß-Glc |
ß-Glc - a-Rha (2 à
1) |
Steviole, the Controversial Metabolite of Stevioside
|
A mutagenic effect of stevioside could not conclusively be detected. According to Pezzuto et al. such an effect of steviole – the aglycone and metabolic waste product of stevioside – was observed in experiments using Salmonella typhimurium, but only occurred after metabolic activation. Such an effect was ruled out for all other constituents and waste products of the sweetener. [7]
|
| Fig.2: Stevia rebaudiana Bertoni (Suttajit et al.) |
Further experiments with Salmonella typhimurium, Escherichia Coli and Bacillus subtilis failed to proof such a mutagenity. The direct mutagenic effect of 15-oxo-steviole continued to be proved and later refuted by various authors. Those studies providing evidence for the mutagenic effect could only prove a very slight impact of steviole. One problem is that the production of steviole as such has not been satisfactorily shown to happen at the point of digestion. Some authors assume that even if such a metabolic mechanism existed, the use as sweetener was unobjectionable. Furthermore, the possibility of toxic effects being caused by impurities in the sweetener itself cannot be neglected. Hamsters turned out to be the most sensitive laboratory animal to stevioside. Metabolites built up which so far could not be identified in rats nor humans. Hence, it remains to be doubted whether studies with hamsters concerning stevia are valid. [8]
Intake and Metabolism of Stevioside
Geuns et al. showed that stevioside is not taken in by the human body and that even in the case of an intake, this intake is negligible. Neither humans nor various animals produce an enzyme capable of metabolizing stevioside. [9]
Koyama et al.’s experiments with rats and hamsters have shown that certain bacteria native to the appendix are capable of converting stevioside into steviole. Steviole was found in the blood of these animals. [10]
Geuns et al., on the other hand, proved stevioside to be quickly excreted by the body – usually without any change to its structure. [11]
In variance with these results, Geuns et al. was also able to prove a complete metabolism of stevioside in pigs without finding any traces of either stevioside nor steviole in the blood. [12]
Geuns concludes from all these experiments that it is only due to certain bacteria that Stevia can be metabolized. [13]
Stevioside and Carcinogenicity
As already mentioned, Pezzuto observed a mutagenic effect with Salmonella typhimurium (using 90% pure stevioside). These results as such may not be read as proof for a carcinogenic quality of this sweetener, even if bacteria happen to metabolize stevioside into steviole.
On the contrary, several authors consider the oral intake of the sweetener to be unobjectionable. Yamada et al. [14], Xili et al. [15], and Toyoda et al. [16] should be mentioned in this particular context.
JECFA (Joint FAO/WHO Expert Committee on Food Additives) of the WHO concluded in 1999, that there are no indications for a carcinogenic potential. [17] |
|
| What a charming smile… |
Fertility and Teratogenicity
As far as effects on fertility are concerned, various studies have come to quite different conclusions in the past. Planas and Kuae observed a reduced birthrate with rats that had been fed with extracts from Stevia. [18]
Further experiments by Shiotsu could not support Planas and Kuae’s results. There was no significant correlation between the general condition, body weight and fertility rate of the animals. [19]
Geuns points out that no other study documented a negative effect on the fertility or reproduction rate of mice, rats or hamsters. [20]
Yamada et al. could find no significant proof for an effect on spermatogenesis. [21]
Melis might have detected reduced fertility with male rats which were fed high doses of stevia extracts. [22] However, the studies of Oliveira-Filho et al. falsified such an effect without doubt when they exposed laboratory animals to extremely high doses of stevia. [23]
A reducing effect on fertility could only be observed in hamsters. However, hamsters have been found to react more sensitively to stevioside and steviole which leads Geuns to question the validity of these studies. [24]

|
Bioavailability of Nutrients in Food
Geuns investigated the growth rate of chicken which were bred to gain weight intensively. Due to their extreme rate of growth these animals are exceedingly sensitive to the slightest change in their nutrition and particularly suitable for such studies. This experiment documented no effect of Stevia on the growth rate of the chicken, leading us to conclude that the intake of essential nutrients such as amino acids, vitamins or minerals remains unaffected by Stevia. Furthermore, Geuns could find no significant correlation between nutrition, weight increase or rate of laying eggs with laying hens. Furthermore, there was no difference in the ratio of egg yolk and egg white. [25]
In reference to numerous similar studies, Geuns concludes that there is not indication for effects on bioavailability or physiology. [26]
|
Precaution and control
is of top priority when working with risk group 2 bacteria. |
Stevioside and Caries
According to Das et al., neither stevioside nor rebaudioside can be said to have cariogenic potential. [27]
Geuns also showed that a high concentration of stevioside impedes the growth of certain types bacteria. However, as Stevia is only used in small quantities its main benefit consists in substituting saccharose, the main cause for caries . [28]
Bibliography 1. Safety evaluation of certain food additives, WHO food additives, Series 54, World Health Organization, Geneva, 2006, S. 117
2. Geuns Jan M.C.: Stevioside.Phytochemistry 2003, 64, 913 – 921
3. Geuns Jan M.C.: Stevioside.Phytochemistry 2003, 64, 913 – 921
4. Xili L., Chengjiany B., Eryi X., Reiming S., Yuengming W., Haodong S., Zhiyian H.: Chronic oral toxicity and carcinogenity study of stevioside in rats. Food Chemical Toxicology 1992, 30, 957-965
5. Toskulkao C., Chaturat L., Temchareon P., Glinsukon T.: Acute toxicity of stevioside, a natural sweetener, and ist metabolite, steviol, in several animal species. Drug Chemical Toxicology 1997, 20, 31-44
6. Geuns Jan M.C.: Stevioside.Phytochemistry 2003, 64, 913 – 921
7. Pezzuto J. M., Compadre C. M., Swanson. S., Dhammika Nanayakkara M. P., Kinghorn D.: Metabolically activated steviol, the aglycone of stevioside, is mutagenic. Medical Sciences 1985, 82, 2478-2482
8. Geuns Jan M.C.: Stevioside.Phytochemistry 2003, 64, 913 – 921
9. Geuns Jan M.C.: Metabolism of stevioside in pigs and intestinal absorption characteristics of Stevioside and rebaudioside A and Steviol. Food Chemical Toxicology 2003, 41, 1599-1607
10. Koyama E., Kitazawa K., Ohori Y., Izawa O., Kakegawa K., Fujino A., Ui M.: In vitro metabolism of the glycosidic sweeteners, stevia mixture and enzymatically modified stevia in human intestinal microflora. Food and Chemical Toxicology 2003, 41, 359-374
11. Geuns, Jan M. C., Bruggemann V., Buyse J.G.: Effect of stevioside and steviol on the developing broiler embryos, Journal of agricultural and food chemistry 2003, 51, 5162-5167
12. Geuns Jan M.C.: Metabolism of stevioside in pigs and intestinal absorption characteristics of Stevioside and rebaudioside A and Steviol. Food Chemical Toxicology 2003, 41, 1599-1607
13. Geuns Jan M.C.: Stevioside.Phytochemistry 2003, 64, 913 – 921
14. Yamada A., Ohgaki S., Noda T., Shimizu M.: Chronic toxicity study of dietary Stevia extracts in F 344 rats. Journal of the Food Hygienic Society of Japan 1985, 26, 169-183
15. Xili L., Chengjiany B., Eryi X., Reiming S., Yuengming W., Haodong S., Zhiyian H.: Chronic oral toxicity and carcinogenity study of stevioside in rats. Food Chemical Toxicology 1992, 30, 957-965
16. Toyoda K., Matsui H., Shoda T., Uneyama C., Takada K., Takahashi M.: Assessment of Carcinogenicity of Stevioside in F344 Rats. Food and Chemical Toxicology 1997, 35, 597-603
17. Safety evaluation of certain food additives, WHO food additives, Series 42, World Health Organization, Geneva, 1999, S. 119-143
18. Planas G.M., Kuae J.: Contraceptive properties of Stevia rebaudiana. Science 1968, 162, 1007
19. Shiotsu S.: Fertility study of Stevia decoction in rats. Technical Journal of Food Chemistry and Chemicals 1996, 4, 108-113
20. Geuns Jan M.C.: Stevioside.Phytochemistry 2003, 64, 913 – 921
21. Yamada A., Ohgaki ., Noda T., Shimizu M.: Chronic toxicity of dietary Stevia Extracts, Journal of the Food Hygienic Society of Japan 1985, 26, 169-183
22. Melis M.S.: Effects of chronic administration of Stevia rebaudiana on fertility in rats, Journal of Ethnopharmacology 1999, 167, 157-161
23. Oliveira-Filho R. M., Uehara O.A., Minett C., Calle L.: Chronic administration of aqueous extract of Stevia rebaudiana Bertoni in rats: endocrine effects, General pharmacology 1989, 20, 187-191
24. Geuns Jan M.C.: Stevioside.Phytochemistry 2003, 64, 913 – 921
25. Geuns J.M.C., Malheiros R.D., Moraes V.M.B., Decupere E.M., Compernolle F., Buyse J.G.: Metabolism by chickens, Journal of agricultural and food chemistry 2003, 51, 1095-1101
26. Geuns Jan M.C.: Stevioside.Phytochemistry 2003, 64, 913 – 921
27. Das S., Das A.K., Murphy R. A., Punwani I.C., Nasution M. P., Kinghorn A. D.: Evaluation of the Cariogenic Potential of the Intense Natural Sweeteners Stevioside and Rebaudioside A, Caries Res. 1992, 26(5), 363-6
38. Geuns Jan M.C.: Stevioside.Phytochemistry 2003, 64, 913 – 921
List of figures
1. Geuns Jan M.C.: Stevioside.Phytochemistry 2003, 64, 913 – 921
2. Suttajit M., U. Vinitketkaumnuen, U. Meevatee, D. Buddhasukh: Mutagenicity and Human Chromosomal Effect of Stevioside, a Sweetener from Stevia rebaudiana Bertoni. Environmental Health Perspectives Supplements. 1993, 101, 53-56. |
